The main purpose of Negative staining is to study the morphological shape, size and arrangement of the bacteria cells that is difficult to stain. eg: Spirilla. It can also be used to stain cells that are too delicate to be heat-fixed.
It is also used to prepare biological samples for electron microscopy. It is used to view viruses, bacteria, bacterial flagella, biological membrane structures and proteins or protein aggregates, which all have a low electron-scattering power. It is also used for the study and identification of aqueous lipid aggregates like lamellar liposomes (le), inverted spherical micelles (M) and inverted hexagonal HII cylindrical (H) phases by Negative staining transmission electron microscopy.
Principle of Negative Staining
Negative staining requires an acidic dye such as India Ink or Nigrosin.
India Ink or Nigrosin is an acidic stain. This means that the stain readily gives up a hydrogen ion (proton) and the chromophore of the dye becomes negatively charged. Since the surface of most bacterial cells is negatively charged, the cell surface repels the stain. The glass of the slide will stain, but the bacterial cells will not. The bacteria will show up as clear spots against a dark background.
Reagents of Negative Staining
India ink
Nigrosin
Nigrosin 100 gm/L, Formalin 5 ml/L in water
Procedure of Negative Staining
1. Place a very small drop (more than a loop full, less than a free falling drop from the dropper) of nigrosin near one end of a well-cleaned and flamed slide.
2. Remove a small amount of the culture from the slant with an inoculating loop and disperse it in the drop of stain without spreading the drop.
 |
3. Use another clean slide to spread the drop of stain containing the organism using the following technique.
 |
4. Rest one end of the clean slide on the center of the slide with the stain. Tilt the clean slide toward the drop forming an acute angle and draw that slide toward the drop until it touches the drop and causes it to spread along the edge of the spreader slide. Maintaining a small acute angle between the slides, push the spreader slide toward the clean end of the slide being stained dragging the drop behind the spreader slide and producing a broad, even, thin smear.
 |  |
5. Allow the smear to dry without heating.
 |
6. Focus a thin area under oil immersion and observe the unstained cells surrounded by the gray stain.
Procedure to view in Transmission Electron Microscope (TEM)
- Hold a coated grid flim side up in a pair of self clamping forceps.
- Make a 1:1 mixture of sample and negative stain (eg. 2% uranyl acetate or 2% sodium or potassium phosphotungstate, pH 7.4). Add 5µl to the grid. Smaller particles adsorb to the grid surface more rapidly than larger particles.
Alternatively the sample mixed with fixative can be added to the grid before subsequent negative staining. - Incubate for 30-90 seconds then remove excess liquid with the torn edge of a
piece of filter paper. - Air dry and examine in the TEM.
Results of Negative Staining
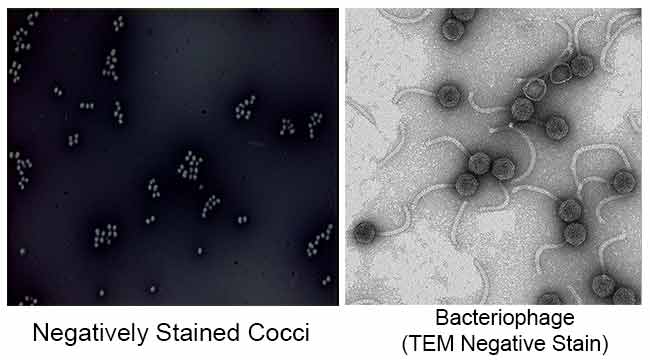
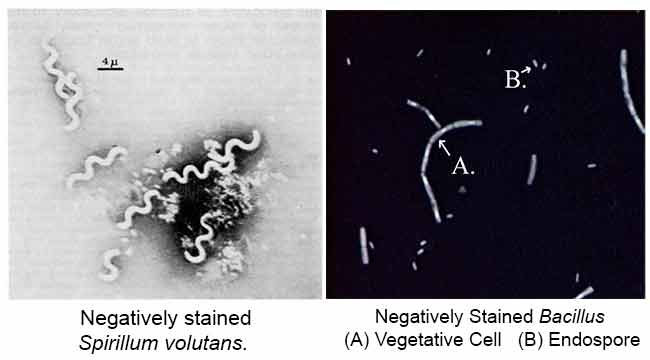
References
- YashRoy R C (1990) Lamellar dispersion and phase separation of chloroplast membrane lipids by negative staining electron microscopy. Journal of Biosciences, vol. 15 (2), pp 93-98.
- Austin Community College
- Western Michigan University
- Los Angeles City College
- Bio-Imaging, SWDSOP, 2004
- Wikipedia

Why do we use a TEM and not any microscope?
simple yet brilliant .. the exact article I was looking for !!
thank you for the info & hope you continue the good work