Sabouraud Dextrose Agar (SDA) is used for the isolation, cultivation, and maintenance of non-pathogenic and pathogenic species of fungi and yeasts. SDA was formulated by Sabouraud in 1892 for culturing dermatophytes. The pH is adjusted to approximately 5.6 in order to enhance the growth of fungi, especially dermatophytes, and to slightly inhibit bacterial growth in clinical specimens.
Composition of SDA
| Ingredients | In gm/L |
|---|---|
| Dextrose (Glucose) | 40 gm |
| Peptone | 10 gm |
| Agar | 15 gm |
| Distilled Water | 1000 ml |
Final pH 5.6 +/- 0.2 at 25ºC.
In addition,
Sabouraud Dextrose Broth is the same formulation as above, without agar added.
Final pH 5.6 +/- 0.2 at 25ºC.
Sabouraud Dextrose Agar with Chloramphenicol contains 50.0 mg of chloramphenicol.
Final pH 5.6 +/- 0.3 at 25ºC.
Sabouraud Dextrose Agar with Chloramphenicol and Gentamicin contains 50.0 mg of chloramphenicol and 5.0 mg gentamicin.
Final pH of 5.6 +/- 0.3 at 25ºC.
Sabouraud Dextrose Agar with Chloramphenicol and Tetracycline contains 50.0 mg of chloramphenicol and 10.0 mg of tetracycline.
Final pH of 5.6 +/- 0.3 at 25ºC.
Sabouraud Dextrose Agar, Emmons has only 20.0 gm of dextrose.
Final pH of 6.9 +/- 0.2 at 25ºC.
Principle of SDA
Peptone (Enzymatic Digest of Casein and Enzymatic Digest of Animal Tissue) provide the nitrogen and vitamin source required for organism growth in SDA. Dextrose is added as the energy and carbon source. Agar is the solidifying agent.
Chloramphenicol and/or tetracycline may be added as broad spectrum antimicrobials to inhibit the growth of a wide range of gram-positive and gram-negative bacteria. Gentamicin is added to further inhibit the growth of gram-negative bacteria.
The neutral pH of the Emmons modification seems to enhance the growth of some pathogenic fungi, such as dermatophytes.
Uses of SDA
- SDA is primarily used for the selective cultivation of yeasts, molds and aciduric bacteria.
- The medium is often used with antibiotics for the isolation of pathogenic fungi from material containing large numbers of other fungi or bacteria.
- This medium is also employed to determine microbial contamination in food, cosmetics, and clinical specimens.
Preparation of SDA
- Combine all ingredients in ~900 ml of deioinized water.
- Adjust to pH 5.6 with hydrochloric acid and adjust final volume to 1 liter.
- Heat to boiling to dissolve the medium completely.
- Autoclave at 121ºC for 15 minutes.
- Cool to ~45 to 50°C and pour into petri dishes or tubes for slants.
Sabouraud agar plates can be inoculated by streaking, as with standard bacteriological media, or by exposing the medium to ambient air. Typically, molds are incubated at room temperature (22 to 25°C) and yeasts are incubated at 28 to 30°C or 37°C if suspected of being dimorphic fungi. Incubation times will vary, from approximately 2 days for the growth of yeast colonies such as Malasezzia, to 2 to 4 weeks for growth of dermatophytes or dimorphic fungi such as Histoplasma capsulatum. Indeed, the incubation time required to acquire fungal growth is one diagnostic indicator used to identify or confirm fungal species.
Result Interpretation on SDA
Identification of fungi is performed by observing various aspects of colony morphology, characteristic microscopic structures, rate of growth, media which supports the organism’s growth, and source of specimen. Yeasts are identified by various biochemical tests.
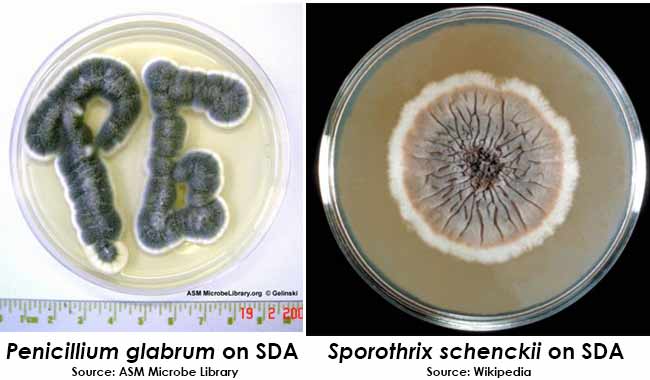
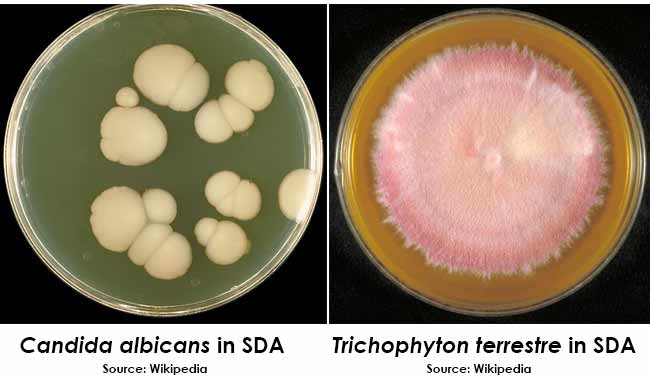
Yeasts will grow as creamy to white colonies. Molds will grow as filamentous colonies of various colors.
Colony Morphology on SDA
Quality Control of SDA
Appearance
Dehydrated medium: Straw colored, free-flowing powder.
Prepared medium: Light straw to straw colored gel.
| Positive controls: | Expected results |
|---|---|
| Candida albicans ATCC® 10231 | Good growth; cream colonies |
| Aspergillus brasiliensis ATCC® 16404 | White mycelium; black spores |
| Negative control: | |
| Uninoculated medium | No change |
Limitations of SDA
- Some strains may be encountered that grow poorly or fail to grow on this medium.
- Antimicrobial agents added into a medium to inhibit bacteria may also inhibit certain pathogenic fungi.
- Avoid overheating a medium with an acidic pH, this may result in a soft medium.
- For identification, organisms must be in pure culture.
- Morphological, biochemical, and/or serological tests should be performed for final identification.
- It does not promote conidiation of filamentous fungi.

What can I use to solidify my Saboraud dextrose broth that will maintain it’s effectiveness and safety? And in what ratio?
I bought broth instead of agar in error
Good afternoon, I am carrying out a research project…aiming at discovering the fungal species in vendored groundnuts from market stalls. I have incubated my samples on Sabouraud Dextrose Agar for three days now. I have noticed some cream colonies, some with raised cream dots, others uniformly cream, others forming strips. Please help me identify the different fungi.
They are bacteria colonia.
While preparing sabouraud dextrose agar with chloramphenicol and gentamicin when to add chloramphenicol and gentamicin?
After sterilization of your media
I’m facing a problem of bacterial growth in sda medium after adding appropriate amount of chloramphenicol. So what should I do?
How we keep SDA plates in cool incubator I mean should keep upsitdown or upside only with suitable reason as well please inform me quickly as soon as possible bcz I have confused about that
Moisture will soon accumulate at the inside part of the lid when a plate is incubated. Hence, it should be placed upside down to avoid contamination from a dripping water which could compromise a culture.
can i release the MLT test SDA plates after 5 dyas icubation if yes please provide the logical resoan
How can I adjust pH of SDA in powder form
Why sometimes SDA media plates don’t solidify after performing MLT? Kindly reply
Not possible or recommended . Why you want to adjust in powder form. Check pH of water used
Always remember, during sterilization pH drops therefore make 0.2 to 0.3 point higher pH than required pH after sterilization because during sterilization 0.2 to 0.3 pH drops
My SDA is coming in at a low pH ~5.2, which is out of range (5.6 ± 2). I’ve tried NaOH. Are there any suggestions for increasing the pH?
NaOH should be enough, 0.1 N should be used. Always remember, during sterilization pH drops therefore make 0.2 to 0.3 point higher pH than required pH after sterilization because during sterilization 0.2 to 0.3 pH drops
Hi. Can I use subouraud dextrose agar as alternative to Potato dextrose agar to isolate fungi such as phytophthora infestans?
Can i use Potato Dextrose Agar as alternative to Sabouraud Dextrose Agar? which is much efficient for isolation of fungi?
SDA plates are incubated invert position it’s correct or wrong method
SDA plates should be incubated upright, do not invert the plates.
Hi, my name is Glory. I’m working on a project in school on the topic ”Micro organisms associated with expired fruit juice”. Please I’ll like to know how to make a gram staining on this test. I’ll also like to know if SDA is important or i should just continue with Nutrient and Simons citrate agar
Why adding glucose in SDA while studying virulent characteristics of Candida spp yet the media really contain dextrose
Apart from penicillium , what other kind of fungi do grow of SDA to give black colonies.?
I was instructed to take the plate home , open for 20 minutes in an area of my home, then close and put in my bag for 2 days. Will something happen to my agar plate if it had been in the car overnight before I was able to start the project?
The temperature it was supposed to be incubated at, might have varied, which might affect the growth. Also, there is a possibility of contamination assuming your plate wasn’t sealed using paraffin tape, etc.
Good morning please I wish to know how many mg of chloramphenicol antibiotic is added to SDA media to prevent bacteria growth.
add 5mg of chloramphenicol into 500ml SDA media.
should this be added as a solution or just as it is, and when should it be added?, i tried adding streptomycin to my SDAY media but it always gets contaminated with bacteria.
How to identify Listeria monocytogen from clinical specimen?
Hello,
Can i use Potato Dextrose Agar as alternative to Sabouraud Dextrose Agar?
Thanks
Hi, i’m helping to give you an answer.
You could use Potato Dextrose Agar as substitution with addition of bacterial antibiotic (such as chloramphenicol) if you want to cultivate fungi.
Hope this help.
should i releas the TYMC plates (SDA Plates after 5 days of incubation.
i need to update in my SOP 5 days incubation for tymc instead of 5-7 days please kindly help me with proper justification
Hi, my name is Helal from Bangladesh. I’ve been working with SDA agar in a food industry for long time,,,,, but still can’t Indentified Yeast growing on this media!!!! Please give some instructions, how to easily Indentified this organism????
You can do a Gram’s stain and observe the hyphae with budding cells. They are Gram positive. You can also do the germ tube test by making a light suspension of 2 large colonies from your SAB agar in 0.5 mls of human or bovine serum. Incubate for 2-3hrs and do a wet mount. You’ll see the hyhae with budding cells. Hope this helps.